 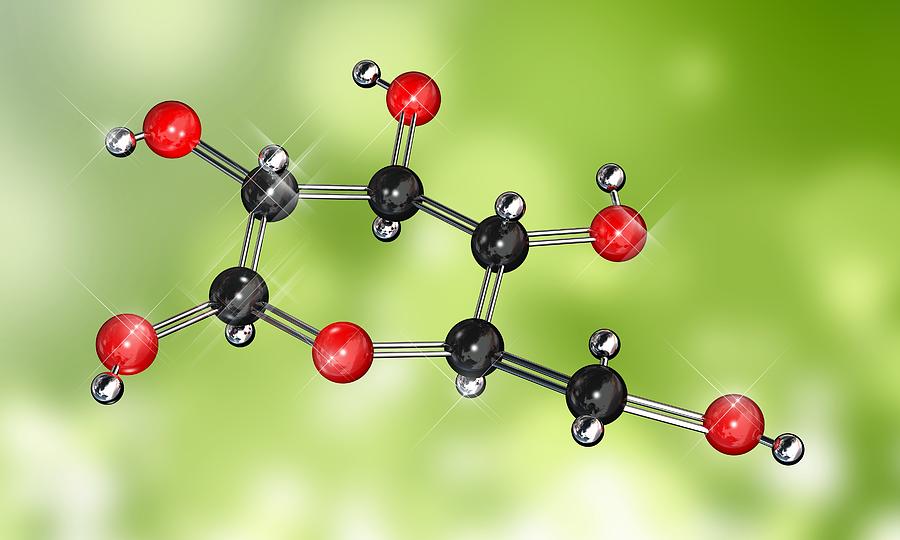
Glucose solutions are used to treat patients with low blood sugar Suppose you prepare a glucose solution using the described procedure. To calculate the number of moles of molecules in 25.4 g of glucose (C6H12O6), we need to first calculate its molar mass. And don’t forget to put the unit g/mol to your final calculated molar mass. Science Chemistry Chemistry questions and answers Glucose (molar mass180.16 g/mol) is a simple, soluble sugar. 
First solve the brackets, then multiplications and at last do the final addition. Always follow the calculation order to avoid any mistakes in calculation.But all these units (i.e g/mol, grams/mole and g/mole) are the same. In some books, you may see the unit of molar mass as grams/mole or g/mole.Step 1: Dissolve 208.7 g of glucose in enough water to make 500.0 mL of solution Step 2. Glucose solutions are used to treat patients with low blood sugar Suppose you prepare a glucose solution using the described procedure. I hope you have understood the short and simple calculation for finding the molar mass of Glucose. Glucose (molar mass180.16 g/mol) is a simple, soluble sugar. Hence the Molar mass of Glucose (C6H12O6) is 180.156 g/mol. So, Molar mass of Glucose (C6H12O6) = Molar mass of 6 Carbon (C) atoms Molar mass of 12 Hydrogen (H) atoms Molar mass of 6 Oxygen (O) atoms. 
Finding molar mass starts with units of grams per mole (g/mol). Convert grams Glucose to moles - Conversion of Measurement Units. You can see that in Glucose (C6H12O6), there are 6 Carbon atoms, 12 Hydrogen atoms and 6 Oxygen atoms. Transcribed Image Text: You are asked to prepare a 1.000L solution of 45 M CHOs (glucose, molar mass 180 16 g/mol) in a lab by dissolving 811.0gof glucose in water Consider the following two scenarios in which you commit a user emor while preparing this solution Prepared in a volumetric flask Prepared in a beaker Assumed volume 1. Do a quick conversion: 1 grams Glucose 0.0055507486072617 mole using the molecular weight calculator and the molar mass of C6H12O6. If they are next to each other, multiply them to get the answer, if one is on top of the other, divide the top one by the bottom one to get the answer.Now, to calculate the molar mass of Glucose, you just have to add the molar mass of all the individual atoms that are present in Glucose. To get the formula from the triangle, cover up the value you want to calculate and use the remaining two values. So, Molar mass of Glucose (C6H12O6) Molar mass of 6 Carbon (C) atoms Molar mass of 12 Hydrogen (H) atoms Molar mass of 6 Oxygen (O) atoms. To remember the formulas in this section, you can use the formula triangles. Fructose, C6H12O6, an isomer of glucose d. Which of the following impurities could account for the low mass percent of carbon in the sample a. For example, about 98.9% of naturally occurring carbon is 12C ('Carbon 12') and about 1.1% is 13C so carbon has an atomic weight of \begin Formula Triangles A chemist analyzes an impure sample of glucose and determines that the mass percent of carbon is 38.2 percent. CH2O is the empirical formula for glucose, C6H12O6 is the molecular formula. The atomic weight (or atomic mass) of an element tells us on average how much one atom of a given element weighs, taking into account typical proportions of isotopes. the percent composition can be used, in conjunction with the molar masses. 
Contents Toggle Main Menu 1 Atomic Weight 2 Molecular Weight 3 Moles 4 Solutions 4.1 1) Molarity 4.2 2) Mass Concentration 4.3 A note on units for concentrations 4.4 Diluting Solutions 5 Test yourself Atomic Weight 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
